New publication published in Microbial Cell Factories
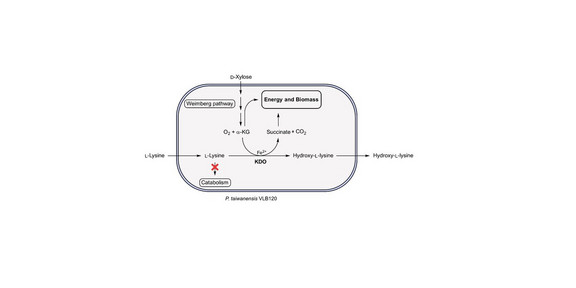
Philipp Nerke and his co-authors establish an efficient whole-cell biocatalyst for the production of hydroxy-L-lysine from L-lysine using D-xylose as the sole carbon source. Hydroxy-L-lysines are valuable chiral building blocks obtained by hydroxylation under catalysis by α-ketoglutarate-dependent lysine dioxygenases (KDOs). To enable a sustainable α-ketoglutarate supply, Pseudomonas taiwanensis VLB120 was modified to provide D-xylose via the Weimberg pathway to α-ketoglutarate for biocatalysis. A mutant strain with inhibited L-lysine catabolism was constructed to prevent substrate degradation of L-lysine, and a library of KDO genes was heterologously expressed. Strains with C4-hydroxylating KDOs achieved efficient production of hydroxy-L-lysine from L-lysine under optimized conditions with elevated L-lysine and Fe²⁺ concentrations. The best performing strain expressing a KDO from Flavobacterium produced approximately 9 g L-¹ hydroxy-L-lysine in a bioreactor. This work provides a new basis for D-xylose-based bioprocesses that can be powered by alternative and renewable carbon sources.